Investing in & steering researchprojects

SUPPORTING DISEASE-MODIFYING RESEARCH PROJECTS
Currently, all available Parkinson's disease treatments are focused on alleviating symptoms i.e. they help to (temporarily) reduce/manage the symptoms of the disease. In order to achieve a real breakthrough, the Demoucelle Parkinson Charity supports research that aims to modify the disease i.e. slow down, halt or even reverse the progression of the disease.
The Demoucelle Parkinson Charity partners with the Michael J. Fox Foundation for Parkinson's Research and applies the following project selection criteria:
— Disease-modifying therapeutics (i.e. focused on finding a cure)
— In pre-clinical development or beyond (i.e. late-stage research)
— In Belgium and elsewhere in Europe
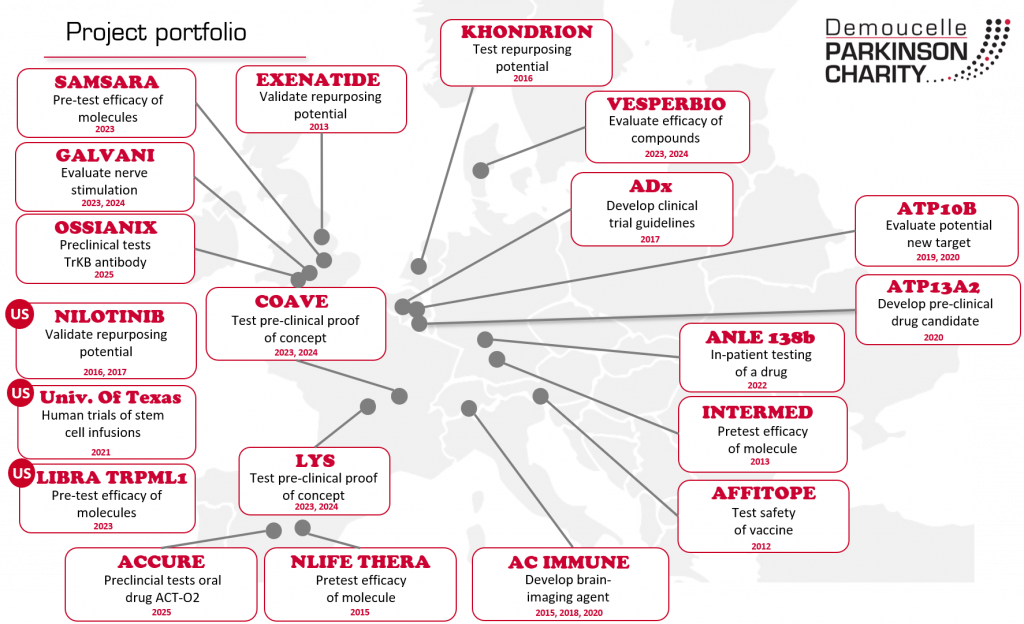
So far, the Demoucelle Parkinson Charity has invested in 18 promising projects. For more information, see below.
INSPIRING THE NEXT GENERATION OF RESEARCHERS
The Demoucelle Parkinson Charity also wants to inspire young researchers in Belgium to pursue their studies and potentially their future careers in investigating Parkinson’s disease. To this end, the charity offers:
- An annual prize for the best masters thesis addressing any aspect of Parkinson’s disease – diagnosis, impact, care, treatment or cure.
-
- Open to all students at Belgian universities and across all disciplines. (Click here for the application rules and process to be followed.)
- Open to all students at Belgian universities and across all disciplines. (Click here for the application rules and process to be followed.)
-
- A PhD fellowship for a doctoral scientist researching an aspect of Parkinson’s disease that could further understanding of the disorder and lead to a cure. Awarded every two years. The next call is foreseen for 2025.
Steering funded projects
We support exciting and promising disease-modifying projects in Europe in collaboration with the Michael J. Fox Foundation.
- InterMed
(DPC invested in 2012).
Objective: identify the potential of a molecule to be neuroprotective and become a disease-modifying therapeutic. Read more
- Affiris: The Affitope Parkinson Vaccine
(DPC invested in 2012)
Objective: perform first phase clinical trial and check impact of the vaccine on α-synuclein, a brain component that plays a central role in the pathogenesis of Parkinson's Disease. Watch our video Read more - Exenatide
(DPC invested in 2013)
Objective: perform a randomized, double-blind, placebo-controlled, proof-of-concept trial to confirm whether the preliminary findings of beneficial effects seen in both motor and non-motor PD symptoms can be replicated. Read more
- nLife Thera
(DPC invested in 2015)
Objective: characterize the dose and efficacy of a new molecule in a model of Parkinson's disease vs a healthy model. Watch our video Read more.
- AC Immune
(DPC invested in 2015 & 2018)
Objective: develop a brain imaging agent for alpha-synuclein, a key protein involved in PD pathology. Watch our video Read more
- Khondrion
(DPC invested in 2016)
Objective: test the effectiveness of a molecule against the mitochondrial dysfunction, often noted in Parkinson’s Disease. Read more
- Nilotinib
(DPC invested in 2016 & 2017)
Objective: check whether the effectiveness of the medicine, shown in open trials, is confirmed in a large double-blind, placebo-controlled trial. Read more. Further info.
- ADx Neurosciences
(DPC invested in 2017)
Objective: develop guidelines for clinical trials.
- ATP10B
(DPC invested in 2019)
Objective: better understand the mutation of this gene, and its potential impact on Parkinson’s disease. Read more
- ATP13A2
(DPC invested in 2020)
Objective: To develop a pre-clinical drug candidate Read more
- University of Texas
(DPC invested in 2021)
Objective: To carry out human trials to test whether multiple intravenous infusions of mesenchymal stem cells will significantly slow the progression of Parkinson’s disease. Read more
- Modag Anle 138b
(DPC invested in 2022)
Objective: Further in-patient testing of a drug targeting the buildup of protein alpha-synuclein in the brain. Read more
- Libra TRPML1
(DPC invested in 2023)
Objective: improve the ability of the brain cells to deal with old and damaged molecules by activating TRPML1.
Read more
- Samsara
(DPC invested in 2023)
Objective: test the ability of small molecules to reduce the accumulation of alpha-synuclein in the brains of mice. Read more
- VesperBio
(DPC invested in 2023 & 2024)
Objective: test the efficacy of Vesper compounds in three in vivo models. Read more
- Lys Therapeutics
(DPC invested in 2023 & 2024)
Objective: better understand the role of T cell infiltration and provide strong proof of concept data for Glunomab immunotherapy use as a treatment for Parkinson’s. Read more
- Coave Therapeutics
(DPC invested in 2023 & 2024)
Objective: Preclinical proof of concept of TFEB in Parkinson disease. Read more. (Last update autumn 2023)
- Galvani Bioelectronics
(DPC invested in 2023 & 2024)
Objective: test the effect of splenic nerve stimulation to reduce inflammation-driven Parkinson’s progression and LRRK2 mutation-driven Parkinson’s risk. Read more
-
TrkB - Ossianix
(DPC invested in 2025)
Objective: To carry out preclinical tests to establish whether the TrKB antibody is able to reach the brain, protect neurons, and increase dopamine production.
Read more -
ACT-02 - Accure
(DPC invested in 2025)
Objective: To finalise critical safety and pharmacology studies needed to submit an Investigational New Drug (IND) application for oral drug ACT-O2 targeting the PREP protein.
Read more
The Demoucelle Parkinson Charity receives frequent project updates and participates in project check-in calls and in-person project assessments organized by staff of the Michael J. Fox Foundation.
Please click here to read updates on our projects (December 2022).
You want more info? Send us een email (info@demoucelle) and we'll get back to you